PickSuck and TissueCutSure Breast Biopsy System
Vacuum-assisted breast biopsy consumables for precise sampling and pathology-friendly specimens.
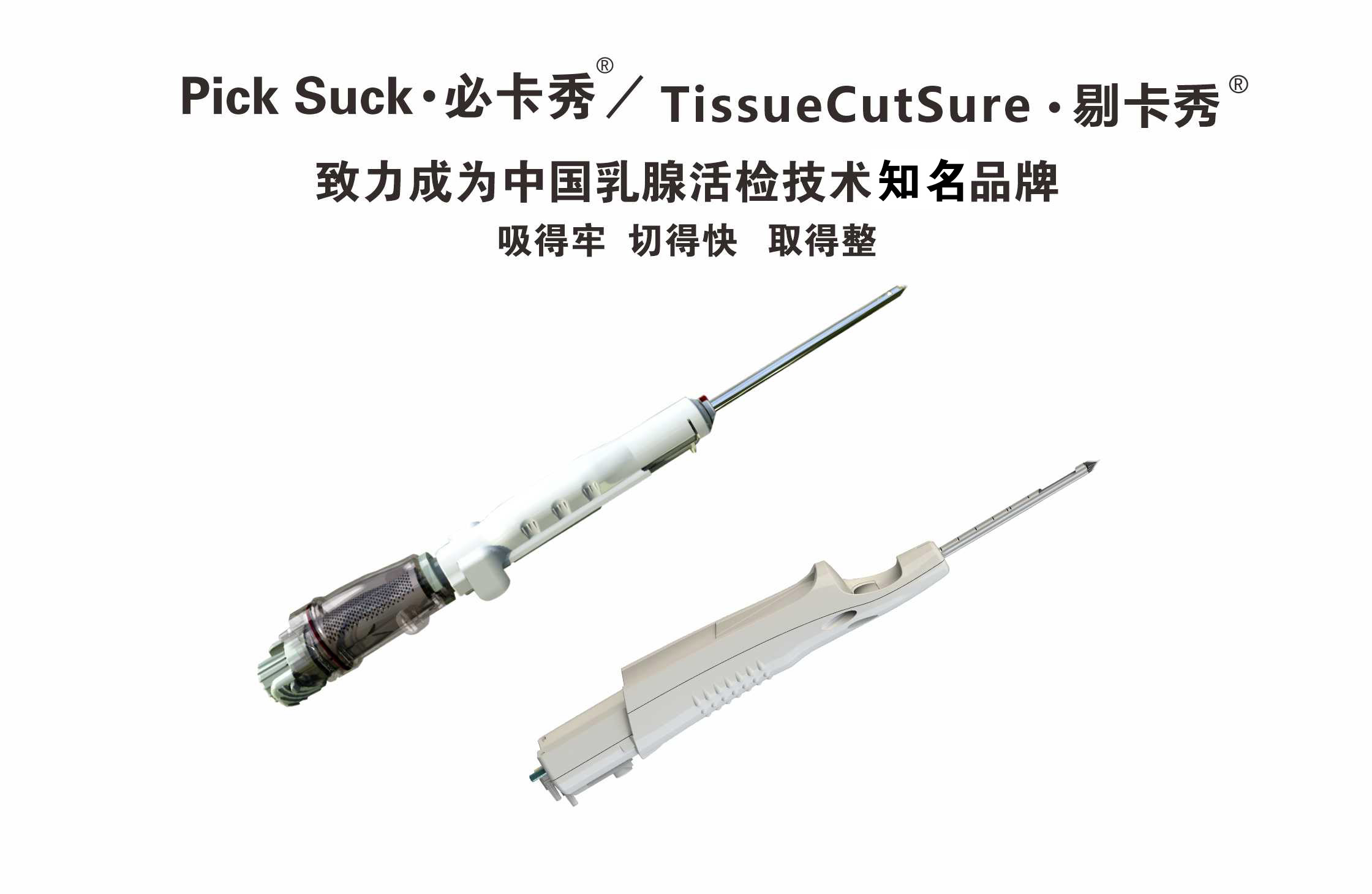
Vacuum-assisted breast biopsy consumables for precise sampling and pathology-friendly specimens.

The following information is provided for professional evaluation. Availability, registration status, and configuration may vary by country.
PickSuck and TissueCutSure form a vacuum-assisted breast biopsy consumable system for ultrasound- or other imaging-guided tissue acquisition. The workflow is designed for hospitals and day-surgery settings that need reliable specimen volume, controlled retrieval, and pathology-friendly cores.


Breast lesion management often requires histology or cytology after imaging characterization. Minimally invasive vacuum-assisted biopsy (VAB) can reduce open surgical biopsies when guidelines support percutaneous sampling under imaging guidance. Disposable needle sets and tubing are intended for single-patient use to support infection control and traceability.
Use only by qualified personnel trained in imaging-guided breast biopsy. Follow institutional protocols for anticoagulation, sterile technique, specimen labeling, and pathology handoff. Contraindications and precautions follow manufacturer IFU and society guidelines applicable in your country.
Manufactured under a medical-device quality system orientation aligned with international market expectations. Marketing claims, CE/registration status, and catalog numbers must be confirmed for your territory before quotation.
This page summarizes positioning for international lead generation. It does not replace the instructions for use, risk management file, or local regulatory labeling.
Our international sales team will respond with pricing, shipping options, and certification support tailored to your market.
Request a Quote