WinRFA Window Ablation System
Visualization-oriented RF ablation system for gynecologic hysteroscopic workflows where clinically indicated.
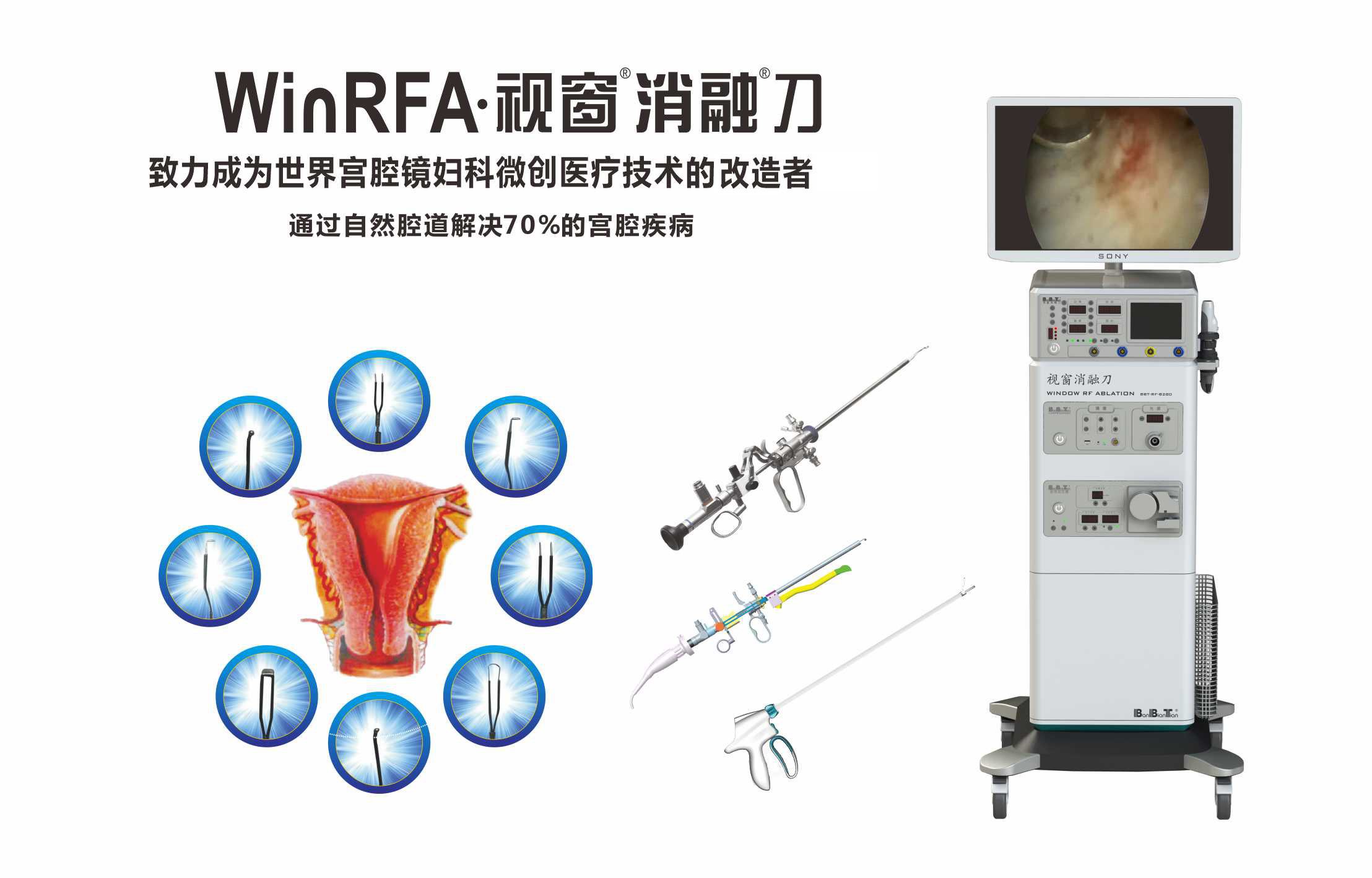
Visualization-oriented RF ablation system for gynecologic hysteroscopic workflows where clinically indicated.

The following information is provided for professional evaluation. Availability, registration status, and configuration may vary by country.
WinRFA is a visualization-oriented RF ablation system for gynecologic hysteroscopic workflows. Marketing materials describe integration of endometrial RF ablation concepts with accessory technologies cleared in your region.

This page summarizes positioning for international lead generation. It does not replace the instructions for use, risk management documentation, or local labeling.
Our international sales team will respond with pricing, shipping options, and certification support tailored to your market.
Request a Quote